An international team of chemists have created a polymer with reversibly changing properties; a plastic with numerous potential applications that is set to make a big impact, specifically in the 3D printing industry.
Such is the scale of the breakthrough that the researchers have included their polymer in a new class of material, which they are calling light-stabilised dynamic materials or LSDMs.
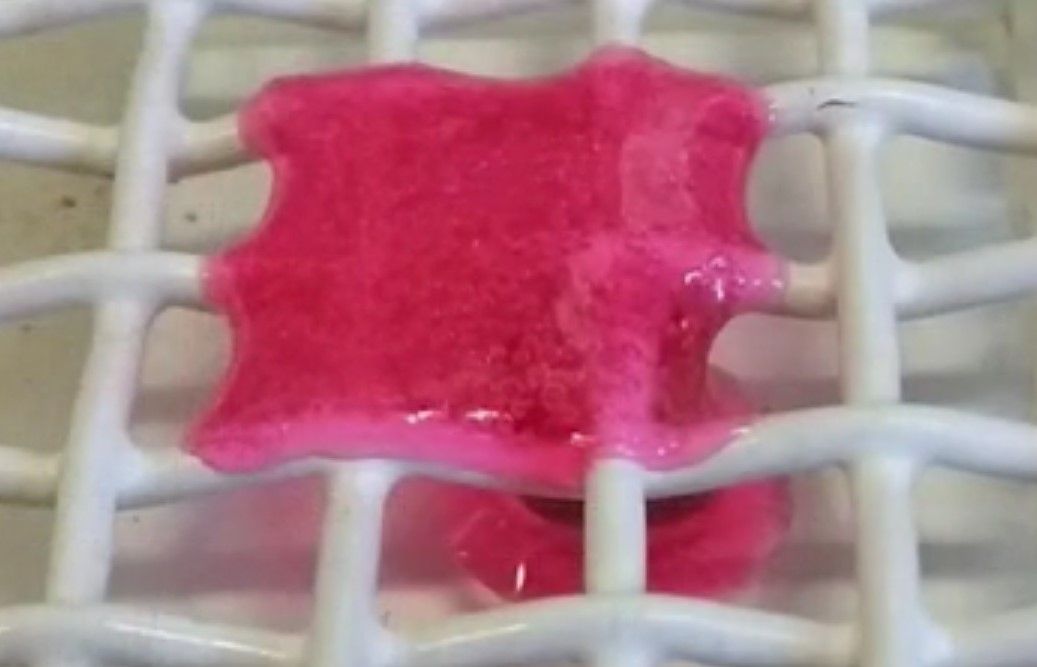
While previous studies have produced dynamic polymers that change structure when a trigger is applied this new material reacts to darkness, in effect when a trigger is removed.
As one of the study’s authors, Professor Barner-Kowollik, explained in an interview with the scientific journal Phys.org, “What makes the discovery unique is that light is used as the trigger to stabilise, rather than destroy, chemical bonds.”

The new polymer was devised during a cooperative work by researchers from Queensland University of Technology (QUT), Ghent University (UGent) and Karlsruhe Institute of Technology (KIT). Together they were able to combine two inexpensive chemical compounds to create a polymer that is stable and strong when a green LED light is shone on it, but melts into a liquid when the light is turned off. Effectively, the main trigger for the polymer is darkness, something which is the opposite of what is usually done in chemistry and that “many people didn't think could be done”.

Amazingly, not only are the raw materials for this new substance relatively cheap, but one of them is the compound naphthalene, a well-known ingredient in moth repellents.
The team have now published their findings in the Journal of the American Chemical Society, where they explain how they utilized, “… the photo-Diels–Alder reaction of [the compound] triazolinedione with [the compound] naphthalene as a dynamic covalent cross-linking platform that enables green light-induced network formation, while the cross-linked material collapses through spontaneous cycloreversion upon standing in the dark at ambient temperature.”

“Typically, you use different wavelengths of light or additional heat or harsh chemicals to break up the polymer molecule chains that form a network structure,” explains Professor Barner-Kowollik. “However, in this case, we used green LED light to stabilise the network. The trigger to break up the network, make it collapse and flow away is darkness. Switch the light back on and the material re-hardens and retains its strength and stability.”

Adding that, “This is what you call an out-of-equilibrium chemical system. The constant energy of the green light keeps the chemical system in this bonded form, pushing it out of its equilibrium. Take away the light, and the system goes back to its relaxed, lowest energy state.”


Already a number of 3D printing companies have contacted the researchers as they are interested in developing the new polymer for industrial use.
The 3D printing industry is big business, as the technology is used to make all manner of intricate and delicate parts and prototypes, especially in the aerospace and automotive industries. However, current technology is limited to what can be printed onto a surface, as the process involves printing layer upon layer of plastic. This prohibits more complicated designs, particularly those that involve bridges, sharply angled structures, or overhangs.
By using the new polymer as a base or support, these shapes could now be possible. As study’s lead author Dr Hannes Houck outlines, “What you need to 3D print something like a bridge is a support scaffold, a second ink that provides that scaffold during printing of the design, but which you can later remove when it is no longer needed. With a light-stabilised dynamic ink used as a scaffold you could 3D print under light, then switch the light off to let the scaffold ink flow away.”

However, the new material could also have a use as a study tool for cell biology, with “… biologists using it as a cell surface support they could alter by light modulation without damaging the cells.”
Following further research, alternative applications could be found in manufacturing or in recycling, while the team are continuing their work to see if other chemical combinations could achieve the same result.
Incredibly, the new polymer has yet to be given a patent, as the team are hoping that the scientific community will help further expand their understanding of how a lack of light can be such a powerful trigger. As Houck explains, “We debated whether to patent the new material, but decided not to wait and to publish the findings to advance knowledge and understanding of the processes involved.”