Researchers at MIT have found an entirely new approach to the production of industrial chemicals.
It is a method that is potentially carbon neutral, produces no toxic chemical waste, and provides basic chemical building blocks for a wide range of consumer products using only electricity and water.
The breakthrough is based on the production of epoxides, a group of chemicals which are used to manufacture a variety of consumer chemical products, such as detergents, polyester clothing, plastics, and pharmaceuticals.
Epoxides are made from olefins, a generic term for a variety of carbon-containing compounds. The raw materials ethene, propene, butenes, and butadiene are all olefins.

However, current industrial processes to convert these chemicals into epoxides are industrially intensive, require large amounts of energy, are environmentally damaging, or produce toxic waste by-products that are expensive to treat and remove.
“We don’t think about the embedded energy and carbon dioxide footprint of a plastic bottle we’re using or the clothing we’re putting on,” says Karthish Manthiram, an assistant professor at MIT’s Department of Chemistry who led the study. “But epoxides are everywhere!”
The result is that epoxide production is a major influencer on climate change. For example, production of the epoxide called ethylene oxide (which among other things is used to produce plastics, solvents, thickeners, and detergents) creates the fifth-largest CO2 footprint of any chemical made today.

This is due to an energy intensive process, as the MIT press release explains, production combines, “… ethylene molecules and oxygen molecules, subject the mixture to high temperatures and pressures, and separate out the ethylene oxide that forms.”
However, the process has several major drawbacks, not least because, “… those ethylene oxide molecules are accompanied by molecules of CO2 — a problem, given the volume of ethylene oxide produced nationwide. In addition, the high temperatures and pressures required are generally produced by burning fossil fuels. And the conditions are so extreme that the reaction must take place in a massive pressure vessel. The capital investment required is high, so epoxides are generally produced in a central location and then transported long distances to the point of consumption.”
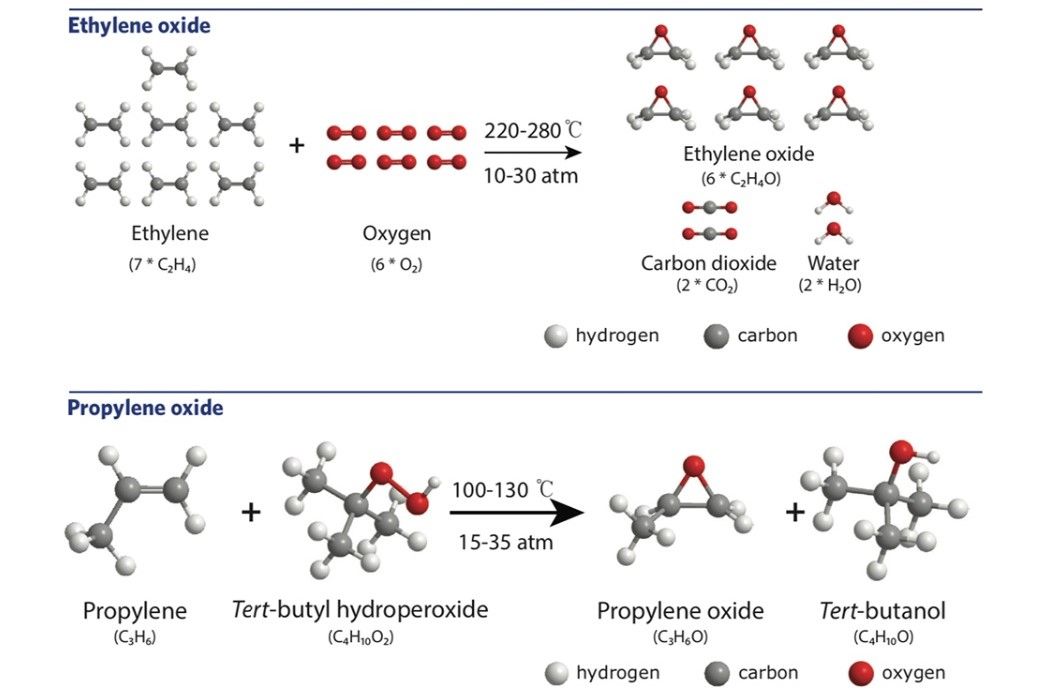
Propylene oxide is another common epoxide that is used to make detergents, polyurethanes, perfumes, and plasticizers. While its production isn’t as energy intensive as ethylene oxide nor does the process produce CO2 it does form a toxic by-product called tert-butyl hydroperoxide which is both highly reactive and flammable with few practical uses.
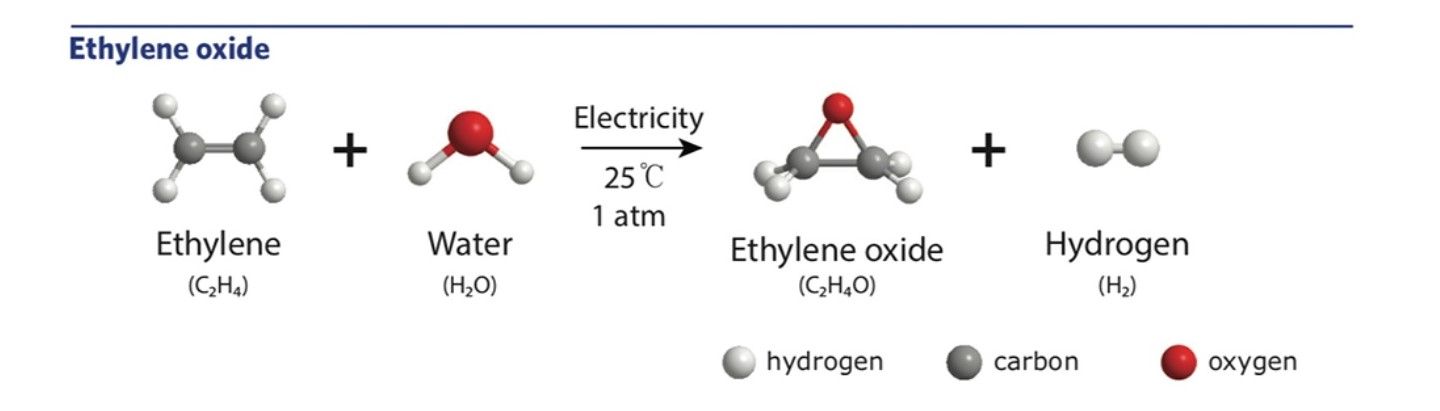
Knowing that the goal in epoxide production is a simple transfer of an oxygen atom from a raw material onto an olefin molecule, Manthiram and his MIT colleagues began to wonder if the oxygen atom could be taken from water.
“Organic chemists would say that it shouldn’t be possible because water and olefins don’t react with one another,” notes Manthiram. “But what if we use electricity to liberate the oxygen atoms in water? Electrochemistry causes interesting things to happen — and it’s at the heart of what our group does.”
And at the heart of electrochemistry is electrolysis: using electricity to split water into oxygen and hydrogen.
Electrolysis is typically used to produce hydrogen gas either for industry or as a fuel, while the oxygen is usually released into the air as an unwanted by-product. However, the team saw the venting of oxygen from this process as a chemical waste stream that could be utilized to make epoxides.
To this end, they proposed using voltage rather than pressure to power the chemical reaction, finding that the conversion of ethylene to ethylene oxide happened at only 0.8 volts.
This was part of the exciting breakthrough; a process to manufacture industrial chemicals at room temperatures with just the power of an AA battery.
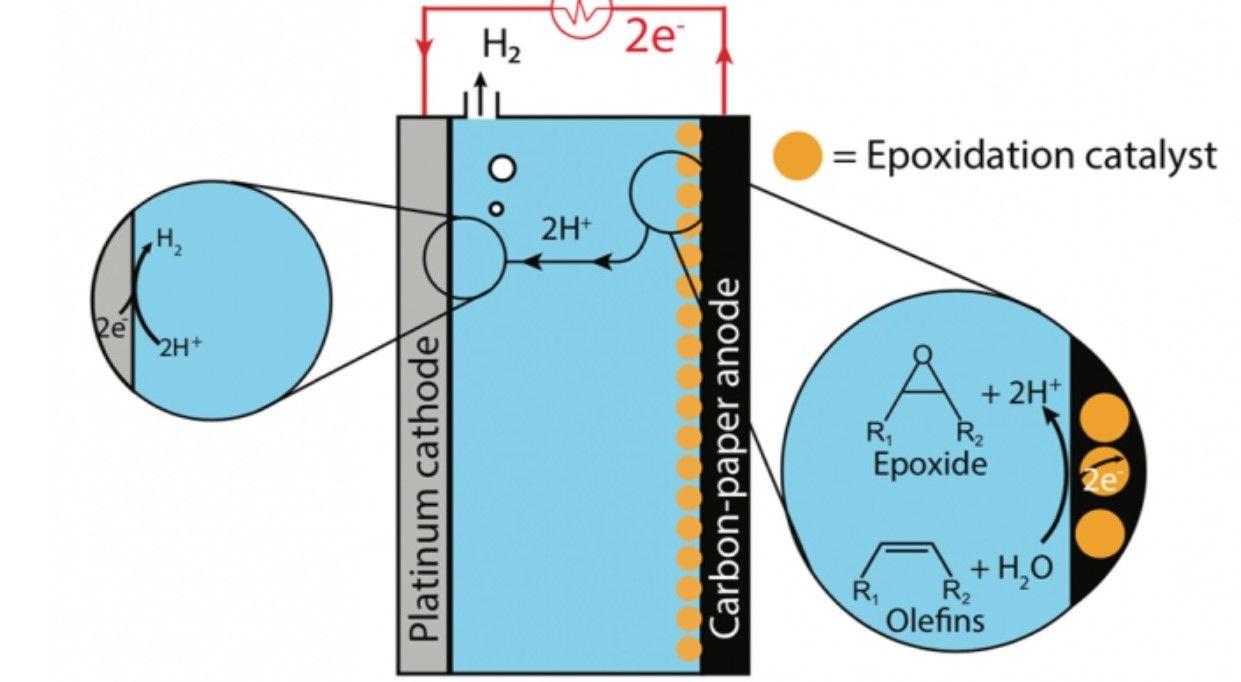
While early theoretical tests were successful, the team did not yet know how quickly the chemical reaction would occur. If it was too slow, then it would not likely be cost-effective. So, the researchers had to design a catalyst – a substance that would speed up the reaction without being consumed.
The team settled on manganese oxide, as it was already known to catalyse the water splitting reaction. They then conducted experiments on an olefin called cyclooctene to test their theory out.
“Ethylene and the like are structurally more important and need to be solved,” explains Manthiram, “but we’re developing a foundation on a well-known molecule just to get us started.”
Results so far have been very promising. As the press release notes, “In one test, the researchers applied 3.8 volts across their mixture at room temperature, and, after four hours, about half of the cyclooctene had converted into its epoxide counterpart, cyclooctene oxide.”
“So, that result confirms that we can split water to make hydrogen and oxygen and then intercept the oxygen atoms, so they move onto the olefin and convert it into an epoxide,” says Manthiram.
Unfortunately, the results also show that the conversion efficiency is only about 30% of what is theoretically possible, which is unlikely to be sufficient to begin industrial production tomorrow. However, this is something the team expected and can explain.
“[It’s] because the electrons are also doing other reactions — maybe making oxygen, for instance, or oxidizing some of the solvent,” says Manthiram. “But for us, 30 percent is a remarkable number for a new reaction that was previously unknown. For that to be the first step, we’re very happy about it.”
Understanding why the low conversion rate exists gives the team hope that they will be able to improve efficiency to a rate that is commercially acceptable.
“Techno-economics will ultimately guide where that number needs to be,” he says. “But I would say that the heart of our discoveries so far is the realization that there is a catalyst that can make this happen. That’s what has opened up everything that we’ve explored since the initial discovery.”
These are still early days with much more work needed on more commercially practical chemical products before thoughts on upscaling can begin. However, Manthiram believes that this work is just ‘the tip of the iceberg’ for a better way to produce epoxides.
Truly, the benefits of this discovery could be far reaching, creating a cheaper alternative to a key industrial chemical process. This is especially true given that the process can also be carried out a smaller scale in locations where the end product is needed (not where fossil fuels are extracted). The process could even be carbon neutral.
As Manthiram states, “If we have solar panels and wind turbines everywhere, why not do chemical synthesis close to where the power is generated, and make commercial products close to the communities that need them?”
This could be a real possibility, especially as the process requires no huge and expensive pressure tanks and produces no harmful or toxic by-products. Localized production would also reduce transportation costs.
“The need to reduce CO2 emissions and energy use is already pushing research on chemical manufacturing towards using electricity from renewable sources,” states the MIT press release.
These are dynamic times for chemical production and this discovery could help lead the way. As the report concludes, “… the face of the chemical industry will change dramatically in the years to come.”
Photo credit: MIT, marcinjozwiak from Pixabay, Michael Gaida from Pixabay, & マクフライ 腰抜けfrom Pixabay