Modern life is digital.
This means that today, cars have more computing power than Apollo 11. What was once a telephone is now a small computer and the Internet of Things will soon have a CPU in the fridge communicating with your laptop to confirm a shopping list.
A world full of computing power (with so much of it portable) requires a lot of electricity and a improved battery systems.

Fortunately, lithium-ion battery production is meeting demand, creating devices that can hold a charge and work when needed. However, as demand begins to grow, with larger, more powerful mobiles or devices that require ultra-light, ultra-small batteries, the limits of lithium-ion battery technology are being reached.
Some of the biggest limits on lithium-ion battery performance include:
· Aging through use – Lithium-ion batteries perform best when they have been charged and discharged for less than 500 cycles. Beyond this they may hold less and less charge.
· Aging over time– Lithium-ion battery performance suffers as the battery gets older even if not used.
· Protection circuitry - another lithium-ion battery drawback is that they require protection circuitry integrated to ensure they are kept in their safe operating limits.
· Temperature – Lithium-ion batteries can be damaged if stored or used in extremes of hot or cold.
· Travel restrictions – While the risk of fire is small, a number of very public incidences has led to restrictions on transporting lithium-ion batteries in the hold on planes.
· Cost – Lithium-ion batteries typically cost about 40% more to manufacture than nickel-cadmium cells.
· Power – They are not as powerful as some alternative rechargeable battery designs.
· Charge levels – Lithium-ion batteries can be damaged if overcharged or if the power drains too low.
Solving these drawbacks is one of the key roles that nanomaterial researchers have set themselves. A topic of research that aims to improve the rechargeable battery market and help meet the insatiable demand for batteries that can hold more power for longer and can last for more cycles than ever before.
Key to this is understanding how current electrode materials can expand significantly as they hold a charge. The constant expansion at full charge and retraction at each discharge causes mechanical problems, such as cracking, at the electrode surface. This issue, combined with low electrical, thermal, and ionic conductivity, all shorten the life span and ability to charge in many batteries.
Lithium-ion batteries also suffer from the formation of dendrites, which can lead to safety as well as lifecycle issues.
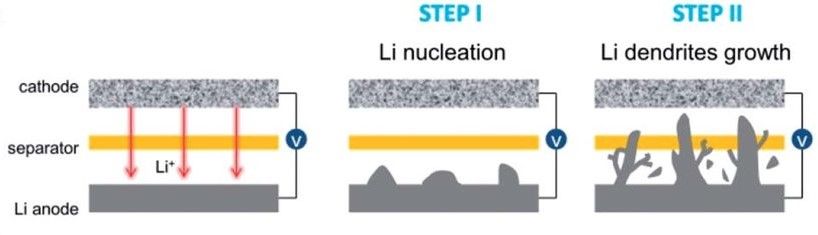
Many of these problems can be solved by applying carbon nanotubes (CNTs) at the surface of the electrode. Alternatively, the carbon nanotubes can be constructed to form a framework into which a lithium insertion material is added.
Carbon nanotubes are superb conductors of electricity, and due to their incredibly small size it is possible to arrange 100 billion nanotubes on a square centimetre of electrode. This means that the nanotubes are able to provide billions of connections at the surface of the electrode, creating a battery with five times longevity, three times the storage capacity, and ten times the power. Charging times have also been reduced to just a few minutes.

It is because of this that CNTs have been at the heart of material research and design for the past two decades. A situation that has not only improved material design in lithium batteries, but also other commercial products. It is a branch of research which offers practical solutions to manufacturers in a wide range of industries, such as pharmaceutical products, construction chemicals, polymers, sports equipment, and electronics, as well as countless composites for the automobile and aerospace industries.
Such is the range of applications for carbon nanotubes, that manufacturers can now custom design their own nanotechnologies to provide their products with desired properties. For example, applying CNTs to a material can provide:
· Increased strength
· Increased rigidity of flexibility
· UV protection
· Thermal conductivity
· Chemical resistance
· Antibacterial properties
· Scratch resistance
· Impact strength
· Electromagnetic properties
· Electrical conductivity
As well as providing these unique selling points, the use of CNTs may also reduce raw material costs, as the carbon nanotubes can replace other additives, with their low concentration a potentially easy alternative to bulk ingredients.
One company that arranges nanotechnology for industry are the nanomaterial specialists at NANO CHEMI GROUP.
They have a great deal of experience in supplying manufacturers with processes to apply nanomaterials to improve specific products, and already have patents pending on nanotechnologies for plastic modification.

Their off-the-shelf product range includes solutions for improving rubbers, coatings, PVC, concrete, and plastic granulate. While they are in close cooperation with nanomaterial scientists from Germany, the Ukraine, Czechia, and Luxembourg.
NANO CHEMI GROUP (a subsidiary of AG CHEMI GROUP who sponsor this website) are also constructing a nanomaterial research centre focused solely on finding solutions for industry. At a total cost of more than €1.3 million, the laboratory will be fitted with cutting-edge equipment, such as nanomaterial analysers, microscopes, mixers, milling machines, and devices for measuring the chemical, mechanical, and rheological properties of resins and plastics.

Like many everyday products, battery design has reached its limit using standard raw materials. By reaching out to nanotechnology specialists, battery manufacturers are revolutionising their industry and finding ways to improve to meet their customer demands.
Beyond batteries, the manufacturers of polycarbonate sheeting, synthetic fabrics, epoxy flooring systems, and technical work surfaces are all making better products, increasing sales, and in some cases even reducing raw material costs thanks to nanotechnology.
To find out how nanotechnology can aid your business, visit: AG CHEMI GROUP or NANO CHEMI GROUP.
Photo credit: ThisIsEngineering from Pexels, Tyler Lastovich, freepik, Tower Electric Bikes, & researchgate