With scientists predicting further increases in greenhouse gas emissions and as the Northern hemisphere experienced another mild winter, politicians and the public continue to ponder the climate change challenges that we are all facing. Yet few have considered the role to limiting carbon emissions played by potassium hydroxide.
This impact is possible through potassium hydroxide’s use as a catalyst in the production of biofuel – most notably biodiesel.
Increasing the use of biodiesel would have a significant role in reducing emissions and lowering human impact on climate change. As a December 2012 report notes, “The U.S. Energy Information Administration (EIA) estimates that in 2020, diesel (distillate) fuel consumption in the U.S. transportation sector resulted in the emission of about 432 million metric tons of carbon dioxide (CO2), a greenhouse gas. This amount was equal to about 26% of total U.S. transportation sector CO2 emissions and equal to about 9% of total U.S. energy-related CO2 emissions in 2020.”

Potassium Hydroxide’s Role in Biofuel
The key to potassium hydroxide’s use in making biofuel is the way that it dissolves triglycerides (fats) allowing an alcohol to bond with the resulting fatty acids. It is a process called transesterification which converts fats and oils into biodiesel and glycerine.
Simply put, the starting raw material is a short-chain alcohol. Methanol is the most common alcohol used as it is inexpensive, although ethanol is also popular, and butanol and amylic alcohol are also viable feedstocks.
As an example, about 10 litres of the alcohol is slowly mixed with potassium hydroxide as a catalyst (although sodium hydroxide [NaOH] may also be used). It is important to ensure that the alcohol is pure with all water removed to improve the efficiency of the ensuing chemical reaction.
To this alcohol-catalyst mixture is added 100 litres of oil (most biofuels are produced from vegetable oil).
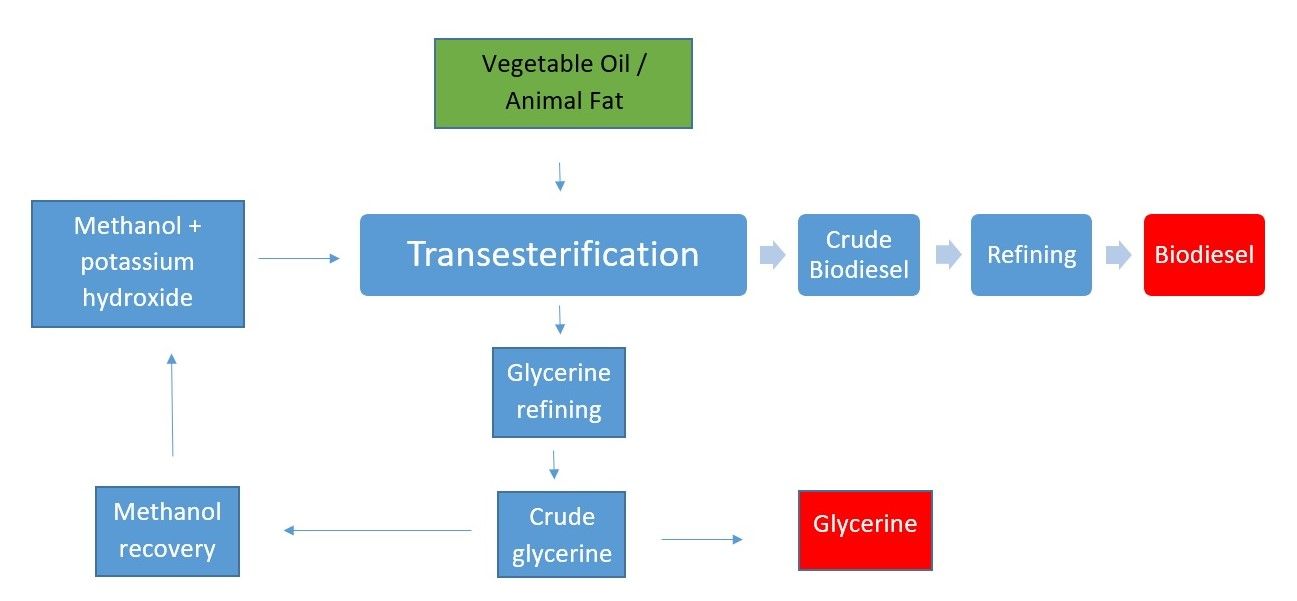
The resulting chemical reaction converts esters (oils and fats) into a mixture of esters of the fatty acids that makes up the oil (or fat). The alcohol then purifies the fatty acids into a biodiesel.
As the boiling point of methanol is approximately 68oC, the transesterification reaction is conducted at atmospheric pressure somewhere between 50 and 60oC.
After the reaction is complete the solution is left to set for a few hours, before the resulting 100 litres of biodiesel is ready for sale/use. Beneath the biodiesel is 10 litres of a sugary co-product called glycerine.
Glycerine (also called glycerol) is widely used in the manufacture of pharmaceuticals and cosmetics.
KOH is highly hygroscopic, meaning that it rapidly absorbs moisture from the air which can restrict the effectiveness of the process. To avoid this, the Super-critical Methanol Production method has been developed which can stop water from contaminating the process with the use of higher temperatures and pressure.
The Advantages of Biodiesel
Biodiesel is a carbon zero product, as any CO2 emitted from burning is equal to the amount absorbed when it was produced. Its plant-based origins means that it is a renewable, clean burning energy source that is suitable for use in existing diesel engines – either as a blend with fossil-fuel diesel or in a pure form.
As well as lowering carbon emissions, engines running on biodiesel also significantly reduce particulate emissions, helping to keep the localized air clean.

Of course, the switch from fossil fuels to biofuels will not happen overnight. Supply chains for products such as KOH will need to be strengthened and a huge investment into biofuel production will be required.
Additionally, more agricultural land will need to be set aside for growing crops that can be used in the biofuel production process. This may meet with political and popular resistance as food prices have risen considerably over the last year or so. The typical feedstocks used in biofuel production are rapeseed (in Europe), soybean (in Argentina and the U.S.), palm (in Asia and Central America) and sunflower oil.
Agricultural land may be kept for food by adopting more used vegetable oils, waste yellow grease, or unwanted animal fats which can be recycled for use in the production of biofuel.

Reducing the amount of carbon produced by the transport sector will not be easy, but it is very possible with biodiesel and potassium hydroxide playing a part in making the change.
As a November 2021 report by European Environment Agency notes, “Between 2010 and 2019, [greenhouse gas] emission intensity [of fuels sold for road transport] decreased by 4.3%, mostly due to the increased use of biofuels.” While the U.S. Environmental Protection Agency has stated that advanced biofuels, “are capable of reducing greenhouse gas emissions [from transportation] by at least 50 percent.”
We all have a role to play in tackling the issue of climate change, and so do biofuels produced with potassium hydroxide.
Photo credit: Alexander Grishin from Pixabay, Gerd Altman, Khunkorn Laowisit from Pexels, & Tom Fisk